It is only recently that a clear link has been established between stem cells and cancer in a variety of cancer types since cancers often arise from the transformation of normal stem cells. In solid organs, cancer stem cells create all the cells in the tumors. But in blood cancers they create havoc in the circulatory and immune systems by spawning large numbers of immature or abnormal cells (usually white blood cells).
For example, in leukemia, these abnormal cells pool in the bone marrow and displace healthy cells, attack or overwhelm the immune system, or cause a platelet or red blood cell deficiency leading to poor wound healing or anemia. Because a stem cell always retains a “copy” of itself in the process of cell division, the leukemia stem cell has a limitless capacity to sustain the disease process. The researchers have identified a type of leukemia stem cell and uncovered the molecular and genetic mechanisms that cause normal blood stem cells to become cancerous.
Leukemic stem cells: where do they come from?
Identification of murine and human acute myeloid leukemia stem cells.
The leukemic stem cell niche: current concepts and therapeutic opportunities.
However, some types of cancers may not originate from stem cells as previously believed.
Traditional chemo and radiation treatments target fast-dividing cells. Cancer stem cells (CSCs) divide slowly. Chemo and radiation doesn’t kill them, and they live on to make new tumors in close-by and distant places. This helps to explain why cancer and leukemia sometimes return after a remission – the seeds of the disease are in these special cancer stem cells that are by nature self-replicating.
Fear of recurrence is common among survivors. Although having some concerns about recurrence is natural, too much worrying can affect your quality of life. Remission of cancer can be temporary or permanent. If remission is temporary, cancer recurs (relapses) either in the same place where the disease first began (primary site) or in different places in the body (metastases). Recurrence rates from different cancers can vary widely, from 5 to 95 percent depending on how far the original tumor had spread, its particular molecular characteristics and other clinical factors. But what makes cancer come back?
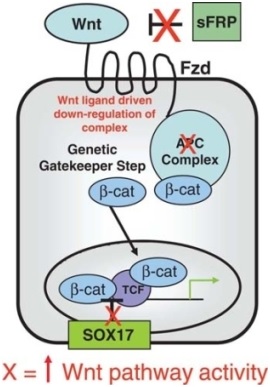 Wnt is one of those biochemical pathways that are thought to be involved in the occurrence of cancer stem cell (CSC). Researchers have identified 19 genes in the Wnt gene family. These genes provide instructions for making similar proteins that participate in chemical signaling pathways in the body. Some Wnt proteins are specific to certain cells and tissues.
Wnt is one of those biochemical pathways that are thought to be involved in the occurrence of cancer stem cell (CSC). Researchers have identified 19 genes in the Wnt gene family. These genes provide instructions for making similar proteins that participate in chemical signaling pathways in the body. Some Wnt proteins are specific to certain cells and tissues.
Studies have demonstrated that by turning off the Wnt signalling pathway, which is constitutively active in colorectal cancer cells, they could trigger in vitro the differentiation of the transformed colon cancer cells into differentiated, non-proliferative, colonic cell types. This shows that the Wnt signal is a “master switch” for the control of colon cancer and that it is possible to knock cancer cells off their proliferative “vicious” circle and trigger their differentiation leading to a non-cancerous phenotype.
Wnt is secreted by cells. Wnt proteins are ligands, which means they can attach (bind) to other proteins called receptors. The free Wnt molecule binds it’s receptor on the cell membrane and activates a survival/growth pathway. Wnt activates a protein called beta-catenin, and this is thought to contribute to the progression of many cancers. Beta-catenin rescues malignant cells that have detached from the original epithelial layer from demise by reducing levels of a cell death-inducing protein. sFRP1 (Secreted Frizzled Protein) is a regulator of Wnt. When Wnt binds to sFRP, it cannot activate the Wnt pathway. Study showed that sFRP is lost in colorectal and breast cancer. SOX17 is most recently defined gene for regulation of Wnt pathway activity found to be virtually, universally, DNA hypermethylated in benign colon polyps. Loss of function for this gene is a vivid example for an epigenetically mediated abnormality in stem/progenitor cell related control.
Stem cells, cancer, and epigenetics.
Mammary stem cells, self-renewal pathways, and carcinogenesis.
Mammary development, carcinomas and progesterone: role of Wnt signalling.
WNT signaling pathway and stem cell signaling network.
Networking of WNT, FGF, Notch, BMP, and Hedgehog signaling pathways during carcinogenesis.
Crypt stem cells as the cells-of-origin of intestinal cancer.
Wnt signaling, stem cells, and the cellular origin of breast cancer.
Wnt/beta-catenin signaling: components, mechanisms, and diseases.
Wnt signaling regulates expression of the receptor tyrosine kinase met in colorectal cancer.
The beta-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells.
APC shuttling to the membrane, nucleus and beyond.
A Wnt-ow of opportunity: targeting the Wnt/beta-catenin pathway in breast cancer.
There is more to learn about the association between Wnt, stemness and the tumour microenvironment.The Wnt signalling pathway is deregulated in most colon cancers; however, only a small proportion of colon cancer cells with Wnt-activating mutations have stem cell-like properties.
Recent study shows that increased activation of Wnt signalling is regulated by factors secreted from myofibroblasts in the stroma surrounding the colorectal tumours. Myofibroblast is a cell that is in between a fibroblast and a smooth muscle cell in differentiation. Tumors are highly complex tissues composed of neoplastic cells and, in the case of cancers, stromal cell compartments containing a variety of mesenchymal cells. Fibroblasts and myofibroblasts often represent the majority of the stromal cells within various types of human cancers.The researchers found that stemness of colon cancer cells is in part orchestrated by the microenvironment and is a much more dynamic quality than previously expected that can be defined by high Wnt activity.
Wnt activity defines colon cancer stem cells and is regulated by the microenvironment.
http://basicmed.med.ncku.edu.tw/admin/up_img/991029-3.pdf
Stromal fibroblasts in cancer: a novel tumor-promoting cell type.
Colon cancer stem cells: promise of targeted therapy.
Thiazolidinedione (TZD) drugs are used widely to treat type 2 diabetes. TZDs act by binding to PPARs (peroxisome proliferator-activated receptors). PPARs are members of the nuclear hormone receptor super-family, regulating gene expression via their ligand-activated transcriptional activity. The ligands for these receptors are free fatty acids (FFAs) and eicosanoids. When activated, the receptor migrates to the DNA, activating transcription of a number of specific genes.TZD downregulate Wnt/beta-catenin signaling pathway.
Quercetin, vitamin D and curcumin regulate WNT/beta-catenin signaling pathway.
Vitamin D and Wnt/beta-catenin pathway in colon cancer: role and regulation of DICKKOPF genes.
The role of nutraceuticals in the regulation of Wnt and Hedgehog signaling in cancer.