Cancer immunotherapy is the use of the immune system to reject cancer. Gammadelta (γδ) T cells are an immune cell found within epithelial tissues. Epithelial tissue is found throughout the body. It is present in the skin, as well as the covering and lining of organs and internal passageways, such as the gastrointestinal tract. Epithelial tissues are also a prominent component of glands, such as prostate gland. γδ T cells play unique and critical roles in recognition of damage or disease in epithelial tissues and provide a crucial first line of defense.
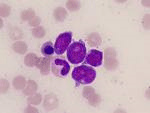 Unlike the alphabeta (αβ) T cells of the immune system (that are launched on a search-and-destroy mission when the skin suffers a cut or other damage), most γδ T …
Unlike the alphabeta (αβ) T cells of the immune system (that are launched on a search-and-destroy mission when the skin suffers a cut or other damage), most γδ T …
Adipose tissue (fat tissue) is currently recognized as an endocrine organ similar to thyroid and pancreas, due to their ability to secrete hormones such as leptin and estrogen and estrogen that could affect metabolism, inflammation and cancer progression. We now know that tumor growth is regulated by interactions between tumor and their tissue microenvironment, such as fibroblasts, immune cells and the extracellular matrix.
Adipose tissue secretes a variety of cytokines, referred to as adipokines. Most adipokines, such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and leptin, are pro-inflammatory. One prominent exception is adiponectin, an anti-inflammatory and anti-cancer adipokine, which promotes insulin sensitization and protects cardiovascular tissue from ischemic injury. Pro-inflammatory adipokines can increase chronic low-grade inflammation in adipose tissue. Tumor-associated adipocytes (fat cells) also contribute to inflammation. Inflamed tumor-associated adipose tissue …
Post-translational modifications such as phosphorylation, methylation, ubiquitination, and acetylation are crucial regulatory modules at the heart of biological processes in the cell and are tightly regulated by a multitude of enzymes. Histones are the chief protein components of chromatin, acting as spools around which DNA winds. The balance of histone acetylation and deacetylation is a critical role in the regulation of gene expression. Histone acetylation induced by histone acetyl transferases (HATs) is associated with gene transcription, while histone hypoacetylation induced by histone deacetylase (HDAC) activity is associated with gene silencing. Unlike acetylation, histone lysine methylation can signal either activation or repression, depending on the site and degree (mono-, di-, or tri-) of methylation.
HDACs also regulate the acetylation status of a variety of other non-histone substrates, including key tumor suppressor proteins …
GSK-3 (glycogen synthase kinase) is a multifunctional serine/threonine kinase that is active all the time in all cells, but particularly highly in the brain. GSK-3 was originally named for its ability to phosphorylate and inactivate glycogen synthase and regulate glucose metabolism. As genes and proteins are discovered, they are often ascribed names based on function—their function at the time of discovery. GSK-3 turned out to be a multifunctional enzyme, able to phosphorylate many proteins. Phosphorylation of the protein acts as a molecular switch, turning the activity on or off. GSK-3 has many phosphorylation targets. It should therefore not be surprising that GSK-3 has both pro- and antiapoptotic roles.
GSK-3 is presently known to be a key regulator of a wide range of cellular functions. GSK-3 regulates numerous cellular processes through …
 Bloodroot (Sanguinaria canadensis) has been used historically by some Native American tribes as a medicinal agent to stimulate the digestive system. It has also been used medicinally as a remedy for fevers, as an expectorant for chronic bronchitis and as a local application in chronic eczema, especially when secondary to varicose ulcers.
Bloodroot (Sanguinaria canadensis) has been used historically by some Native American tribes as a medicinal agent to stimulate the digestive system. It has also been used medicinally as a remedy for fevers, as an expectorant for chronic bronchitis and as a local application in chronic eczema, especially when secondary to varicose ulcers.
Bloodroot is generally prescribed as an external treatment as it is poisonous if ingested in large amounts. In toxic doses, it causes burning in the stomach, intense thirst, vomiting, faintness vertigo, intense prostration with dimness of eyesight. Bloodroot grows primarily in North America and in India. Sanguinarine, the major active compound found in bloodroot, is a very impressive natural medicine. Sanguinarine is a benzophenanthrene alkaloid. It has also been …
Much of the cancer chemopreventive properties of green tea are mediated by EGCG. Cancer cells survive by using multiple pathways. But EGCG is so powerful, it cuts those pathways off. Apparently, EGCG functions as a powerful antiangiogenic and antitumor agent and as a modulator of tumor cell response to chemotherapy. EGCG induces apoptosis and promotes cell growth arrest by altering the expression of cell cycle regulatory proteins, activating killer caspases, and suppressing oncogenic transcription factors and pluripotency maintain factors.
EGCG is also able to induce proteasome inhibition in whole cells. Since the inhibition of the proteasome blocks the activation of NF-kB, it is logical to conclude that proteasome inhibitors such as EGCG would have a strong therapeutic effect against cancer, lymphoma and leukemia….
 EGCG (epigallocatechin gallate) is the most abundant and perhaps most important catechin found in green tea (camellia sinensis). Catechins are polyphenolic antioxidant plant metabolites and belong in the flavonoid family. EGCG functions as a powerful antioxidant, preventing oxidative damage in healthy cells, but also as an antiangiogenic and antitumor agent and as a modulator of tumor cell response to chemotherapy.
EGCG (epigallocatechin gallate) is the most abundant and perhaps most important catechin found in green tea (camellia sinensis). Catechins are polyphenolic antioxidant plant metabolites and belong in the flavonoid family. EGCG functions as a powerful antioxidant, preventing oxidative damage in healthy cells, but also as an antiangiogenic and antitumor agent and as a modulator of tumor cell response to chemotherapy.
There is a huge literature showing that EGCG kills cancer cells of all kinds. EGCG reactivates epigenetically silenced genes in cancer cells and induces apoptosis and promotes cell growth arrest, by altering the expression of cell cycle regulatory proteins, activating killer caspases, and suppressing nuclear factor kappa-B (NF-kB) activation.
The majority of human cancers demonstrate the inactivation of the p53 pathway. p53 …
Metabolic abnormalities of tumor cells offer opportunities of therapeutic targeting. Tumor cells are more sensitive to methionine restriction than normal tissues, a phenomenon known as methionine auxotrophy. Methionine is one of the essential amino acids with many key roles in mammalian metabolism such as protein synthesis, methylation of DNA and polyamine synthesis but cannot be produced in the body, and so must be provided through our diet.
Many cancer cells and primary tumors have absolute requirements for methionine. Methionine-dependent increase in tumor cells is a specific metabolic defect. The biochemical mechanism for methionine dependency has been studied extensively, but the fundamental mechanism remains unclear. Methionine starvation can powerfully modulate DNA methylation, cell cycle transition, polyamines and antioxidant synthesis of tumor cells. Therefore, low methionine in the diet may be an important …
 Bitter melon is a member of the Cucurbitaceae family (cucumbers, pumpkins and watermelons). The bitter melon fruit, sometimes called bitter gourd or wild cucumber, grows in tropical and sub-tropical climates. It is very nutritious vegetable having high therapeutic value. It is used both as food and in medicine to treat type2 diabetes, cancer, infections, HIV (AIDS), menstrual disorder and immune disorders.
Bitter melon is a member of the Cucurbitaceae family (cucumbers, pumpkins and watermelons). The bitter melon fruit, sometimes called bitter gourd or wild cucumber, grows in tropical and sub-tropical climates. It is very nutritious vegetable having high therapeutic value. It is used both as food and in medicine to treat type2 diabetes, cancer, infections, HIV (AIDS), menstrual disorder and immune disorders.
Chemical constituents from whole plants, fruits, and seeds of bitter melon (Momordica charantia) have been isolated and described as following:
Glycosides: momordin, charantin
Alkaloids: momordicin
Others: polypeptide-P
Oils (seed only): stearic, linoleic, oleic acids
Glycoproteins: alpha-momorcharin, beta-momorcharin, lectins
Others: vicine (pyrimidine nucleoside), protein MAP30
Cucurbitane triterpenoids
Previously, the researchers have reported that extract of bitter melon has sugar- and fat-lowering properties. The compounds present in …
Signal transducer and activator of transcription-3 (STAT3) protein is a member of the STAT family of transcription factors which are initially located in the cytoplasm in their inactive form. STAT proteins are part of a group that is sometimes called DNA- binding factors. These proteins bind to DNA sequences, and therefore control the transcription of information from DNA to mRNA. A variety of growth factors and cytokines activate STAT3 by phosphorylating the tyrosine residue in the STAT3 transactivation domain. Phosphorylated STAT3 (p-STAT3) then translocates into the nucleus and induces the expression of a wide variety of target genes involved in tumorigenesis.
Tumor cells depend on STAT3 for continued rapid growth and avoidance of apoptosis. STAT3 is constitutively activated in a wide variety of tumours, including colorectal cancer, liver cancer, breast cancer, …