Millions of people worldwide are affected by Ulcerative Colitis (UC) and Crohn’s Disease (CD), the two most severe digestive afflictions. Both diseases are classified under the medical rubric of IBD (Inflammatory Bowel Disease). Inflammatory mediators have been identified in IBD, and considerable evidence suggests that these mediators play an important role in the pathologic and clinical characteristics of these disorders. The result is an immune response that destroys normal body tissues. However, there is no scientific proof that IBD is an autoimmune disease. It is simply an assumption. The cause of IBD remains poorly understood.
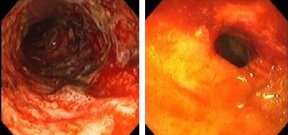 UC and CD share many extraintestinal manifestations, although some of these tend to occur more commonly with either condition. UC and CD cause life impairing symptoms (lots of bloody diarrhea, abdominal pain and weight loss), necessitate long-term dependence on powerful anti-inflammatory/immunosuppressant drugs, and often result in debilitating surgery and even death. UC and CD are likely just two symptoms of the same morbidity rather than two different diseases.
UC and CD share many extraintestinal manifestations, although some of these tend to occur more commonly with either condition. UC and CD cause life impairing symptoms (lots of bloody diarrhea, abdominal pain and weight loss), necessitate long-term dependence on powerful anti-inflammatory/immunosuppressant drugs, and often result in debilitating surgery and even death. UC and CD are likely just two symptoms of the same morbidity rather than two different diseases.
IBD is more common among people of Northern European and Anglo-Saxon origin and occurs more frequently in urban communities than in rural areas. Both UC and CD are more common in white-collar workers. The incidence of IBD has risen with the tide of civilization. These suggest that IBD is due to a combination of factors, including genetic predisposition, environmental factors, including diet, pollution, exposure to industrial chemicals, lack of sunlight exposure (lack of vitamin D hormone), infection and chronic stress, and alterations in the function of the immune system.
Inflammatory bowel symptoms are not a disease of the stomach or intestines. That is just where the symptoms have manifested. The true source of this degenerative disease is imbalanced defense mechanism. Therefore, patients with IBD must correct imbalances in the immune system, hormonal system, digestive system and nervous system first, so that the mucus lining of the gut returns to normal and then the body can absorb nutrients again and thus heal itself.
Epithelial tight junctions in intestinal inflammation.
Do we really understand what the immunological disturbances in inflammatory bowel disease mean?
Intestinal bacteria and inflammatory bowel disease.
Role of reactive metabolites of oxygen and nitrogen in inflammatory bowel disease.
Investigating intestinal inflammation in DSS-induced model of IBD.
 A variety of changes in the gut flora have been proposed as the root cause of IBD. For example, concentrated milk fats, which are abundant in processed and confectionary foods, alter the composition of bacteria in the intestines. These changes can disrupt the delicate truce between the immune system and the complex but largely beneficial mix of bacteria in the intestines. The emergence of harmful bacterial strains in this setting can unleash an unregulated tissue-damaging immune response that can be difficult to switch off.
A variety of changes in the gut flora have been proposed as the root cause of IBD. For example, concentrated milk fats, which are abundant in processed and confectionary foods, alter the composition of bacteria in the intestines. These changes can disrupt the delicate truce between the immune system and the complex but largely beneficial mix of bacteria in the intestines. The emergence of harmful bacterial strains in this setting can unleash an unregulated tissue-damaging immune response that can be difficult to switch off.
The microbial ecosystem in the gut in many different microhabitats can be influenced by diet, leading to formation of metabolic processes that are essential form the bowel metabolism. Current interest therefore focuses on the bacterial community as the source of antigens that fuel the chronic inflammation seen in IBD. We can’t do much about correcting genes that predispose individuals to increased risk for these diseases. However, the balance between host and microbes can be altered back to a healthy state to treat these diseases.
Current concepts of the intestinal microbiota and the pathogenesis of infection.
Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice.
Patients with inflammatory bowel disease exhibit dysregulated responses to microbial DNA.
The gut microbiota and mucosal T cells.
When a person has the wrong species of bacteria and yeasts in the intestines, the condition is called intestinal dysbiosis. Harmful bacteria and fungus spread their toxic humors in the intestines when a natural balance has been disrupted. This can arise in several ways:
High intakes of mono- and disaccharides, and total fats consistently increase the risk developing both forms of IBD. High vegetable intake reduces the risk of UC, whereas increased fruit and/or dietary fiber intake appears protective against CD. One significant change in the modern diet in areas with high incidence of UC is the high intake of sulfur-containing food. Sulfur not recycled by an intestinal sulfur salvage pump passes into the colon where it is metabolized, in some, by sulfatereducing bacteria, a group of bacteria identified as being much more common in individuals with UC and associated with flares of UC, in particular.
Low levels of certain micronutrients, especially vitamin D, may increase the risk of both diseases. Some artificial sweeteners (saccharin and sucralose) also can be one of the causes of IBD. Artificial sweeteners reduce beneficial bacteria, damage the intestine, and cause bacterial, antigen, and particle infiltration.
Role of nutrition and microbiota in susceptibility to inflammatory bowel diseases.
Inflammatory bowel disease: role of diet, microbiota, life style.
Novel role of the vitamin D receptor in maintaining the integrity of the intestinal mucosal barrier.
Etiology of inflammatory bowel disease: a unified hypothesis.
We are exposed to chemicals through the unsafe food we eat, the air we breathe, and the water we drink and bathe in. For example almost all of the dioxin found inside our body got there from eating contaminated food. Whatever its source, somewhere it entered the food chain and made its way into the food we ate. Many seemingly harmless food additives that are in most prepared foods may upset digestion and kill off or damage the friendly intestinal flora. This is especially the case when these are eaten regularly. They often irritate the intestine and alter its pH or other delicate chemical balances in the intestine.
The mucosal epithelium constitutes a physical and functional barrier between the host and components of the external environment, including nutrients, microbes, and toxicants. Recent study shows that mucosal expression of Elafin, a natural protease (digestive enzyme to help digest proteins) inhibitor expressed in healthy intestinal mucosa, is diminished in patients with IBD. Elafin has pleiotropic anti-inflammatory properties.
Interaction between food substances and the intestinal epithelium.
Food-grade bacteria expressing elafin protect against inflammation and restore colon homeostasis.
Therapeutic potential of human elafin.
Modifying the protease, antiprotease pattern by elafin overexpression protects mice from colitis.
Allergic and nutritionally related causes also have been the focus of considerable research. Microparticles, which are part of the concept behind toothpaste as a cause, have been suggested more broadly to be the principal factor initiating IBD. It is confirmed that in UK, about 40 mg of exogenous microparticles are ingested per person per day, through exposure to food additives, pharmaceutical/supplement excipients or toothpaste constituents.
Past and current theories of etiology of IBD: toothpaste, worms, and refrigerators.
The presence of harmful bacteria such as Klebsiella pneumoniae and Proteus mirabilis in the gut correlates with IBD. What are these microbes?
Klebsiella is a type of gram-negative bacteria that can cause different types of infections, including pneumonia, bloodstream infections, wound or surgical site infections, and meningitis. Healthy people usually do not get Klebsiella infections. Klebsiella can be spread through person-to-person contact (for example, from patient to patient via the contaminated hands of healthcare personnel, or other persons) or, by consuming contaminated foods. They are also found in human stool (feces).
Proteus mirabilis is another type of gram-negative bacteria that can be found as part of the micro flora in the human intestine. This organism is not usually a pathogen, but does become a problem when it comes into contact with urea in the urinary tract. From there, infection can spread to other parts of the body. It is one of the species responsible for causing urinary tract infections in thousands of people each year in hospitals. P. mirabilis infection can also lead to the production of kidney and bladder stones
A possible link between Crohn’s disease and ankylosing spondylitis via Klebsiella infections.
The gut flora is profoundly influenced by GMOs (genetically modified organisms). GMO is an organism whose genetic structure has been altered by gene splicing. Farm animals have been raised on GM feed for many years. Certainly it means that ill effects may not show up immediately.
Russian researchers fed Campbell hamsters (which have fast reproduction rates) Monsanto GM soy for two years. It should be noted that hamsters do not evolutionarily eat soy—just as cows fed Monsanto corn are actually ruminants and would not naturally eat corn.
After feeding hamsters for two years over three generations, those on the GM diet, and especially the group on the maximum GM soy diet, showed devastating results. By the third generation, most GM soy-fed hamsters lost the ability to have babies. They also suffered slower growth, and a high mortality rate among the pups. And if this isn’t shocking enough, some in the third generation even had hair growing inside their mouths–a phenomenon rarely seen, but apparently more prevalent among hamsters eating GM soy.
When researchers fed male rats GM soy, their testicles changed from the normal pink to dark blue.
A new example of ectopia: oral hair in some rodent species.
Furthermore, a paper shows that consuming GM corn or soybeans leads to significant organ disruptions in rats and mice, particularly in livers and kidneys. Other organs may be affected too, such as the heart and spleen, or blood cells
http://www.enveurope.com/content/pdf/2190-4715-23-10.pdf
The most obvious nutrition concern of GM foods is the risk of allergic reactions. More than 90% of food allergies occur in response to specific proteins in foods. Allergic reactions to GM foods could lead to problems like leaky gut syndrome. Leaky gut syndrome happens when the intestinal lining becomes inflamed, and the microvilli on the lining become damaged. Leaky gut syndrome is also associated with chronic fatigue and depression.
Furthermore, GM foods actually become part of the bacteria in our digestive tracts and reproduce continuously inside us. Even after we stop eating GM foods, we may still have the GM proteins produced continuously inside us. If the antibiotic gene inserted into most GM crops were to transfer, it could create antibiotic-resistant diseases. Bt toxins (produced from Bacillus thuringiensis bacteria—as a method of natural insect control) inserted into GM crops to kill pests could turn bacteria in our intestines into living pesticide.
Furthermore, animal studies show that DNA in food can travel into organs throughout the body. GM foods genes transferring to our own genes could lead to problems like UC and CD. GM foods could alter our digestive system. The statistical increase in digestive diseases and colorectal cancer can be directly traced to the creation of GM foods. Even after surgery, the UC and CD continued and actually got worse until when to stop eating GM foods and any product containing chemical additives. UC and CD didn’t get there by accident.
Some producers use GM ingredients in their “natural” breakfast cereals. About 1/2 of U.S. corn crop is grown from GMO seeds. Monsanto provides roughly 90% of GMO seeds in the world. Globally, GM is increasingly seen as a trade issue. However, the US wants to protect its biotechnology industries and is aggressively seeking new markets for its export orientated agricultural sector. For example, despite lingering concerns over the safety of GMO for human consumption, the amount of imported GMO corn and soybeans to South Korea totaled 2.05 million tons in 2010, up 92 per cent from 1.07 million tons in the previous year, according to the data by the Ministry for Food, Agriculture, Forestry and Fisheries. Recent reviews from South Korea also report a sharp increase in the incidence and prevalence of IBD. The incidence of pediatric IBD is increasing rapidly as well.
Genetically modified (GM) foods – renewed threat to Europe
Most patients with IBD have no idea that oral health and oral bacterial flora are so important to their treatment. Poor oral health and harmful oral bacteria can lead to intestinal failure, irritable bowel syndrome and other digestive disorders. Harmful oral bacteria can enter your bloodstream and attack other tissues. The connection between systemic health and good oral health is well documented. Maintaining good oral health is one of the most important things that patients with IBD can do. You no longer have to put harmful chemicals, toothpastes and irritants into your mouth (most of which can make your symptoms even worse).
Correlation network analysis applied to complex biofilm communities.
The oral microbiome in health and disease and the potential impact on personalized dental medicine.
Deep sequencing of the oral microbiome reveals signatures of periodontal disease.
Association between Selected Oral Pathogens and Gastric Precancerous Lesions.
If you are what you eat, then patients with UC and CD are toxic dump sites for the dangerous bacteria residues left in the gut by eating most unsafe foods, factory farmed meats and GM crops. Patients with IBD often note significant improvement in their symptoms within three weeks of starting the GinolZym plus Oralzym (OralZym and OralZym-F) therapy. By twelve weeks, the majority are recovering definitively. GinolZym has an inhibitory effect on gram-negative harmful bacteria such as Klebsiella pneumonia.
One twenty six-year-old patient with UC affecting the rectum had daily bloody diarrhea despite medications for years until initiating the Ginolzym plus Oralzym therapy. She took 4 months to become symptom-free. She now maintains her remission with the Ginolzym plus Oralzym therapy. Another patient with CD, After 3 months, he is completely symptom-free without the aid of medications.